 When a voltage is applied across the electrodes, electrons are emitted from the cathode and accelerate towards the anode. The cathode ray tubeĬathode ray tubes, previously known as gas discharge tubes or Crookes tubes, are vacuum glass tubes with two metal electrodes and rarefied gas inside. Thomson in 1897 when he concluded that cathode rays were made up of a previously unidentified negatively charged particle, the electron. The most important use of cathode rays was discovered by J.J. 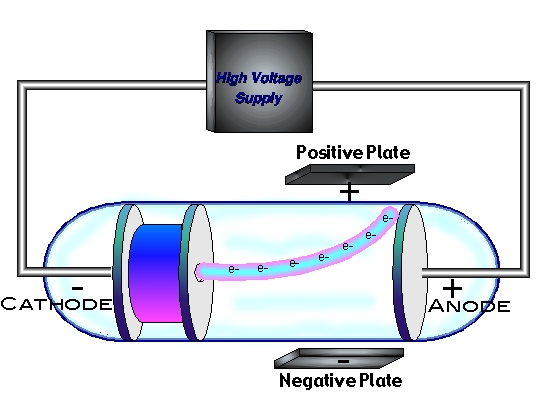
Julius Plücker and Johann Wilhelm Hittorf first observed cathode rays in 1869, and Eugen Goldstein named them in 1876. The properties of cathode rays do not change regardless of the gas used in the tube. Properties of cathode raysĬathode rays are negatively charged particles that travel in a straight route and ionise the gas inside the vacuum tube. The electrode connected to the negative pole of the voltage source is the cathode, and the electrode connected to the positive pole of the voltage source is the anode. To find which electrode is the cathode and the anode, we need to look at the connections between the electrodes and the voltage supply. The electrons emitting from the cathode is what causes this glow. A glow behind the positive electrode (cathode) can be observed when a potential difference (voltage) is applied to the electrodes. 
These discharge tubes are devices that control the electric current between a potential difference applied to the electrodes in a high vacuum. 
If you want to know more about this discovery, then keep reading! What is a cathode ray?Ĭathode rays, also known as electron beams, are streams of electrons detected in discharge tubes (vacuum tubes). These rays played a significant role in discovering the electron. They are also known as e-beam or electron beams. Cathode rays are basically electron streams found in vacuum tubes. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
